|
A nonmetal is typically dull and a poor conductor of electricity and heat. Metals are also malleable (they can be beaten into thin sheets) and ductile (they can be drawn into thin wires). A metal is a substance that is shiny, typically (but not always) silvery in color, and an excellent conductor of electricity and heat. Here no doubt electrons fill in penultimate shell but there is only one electron in valence shell that is 4s.\). In general, atoms are most stable, least. According to the periodic table, oxygen is in group 6 therefore. This outermost shell is known as the valence shell, and the electrons found in it are called valence electrons. We can see from the electron configuration of a carbon atom1 s2 2 s2 2 p2 that it has 4 valence electrons (2 s2 2 p2) and 2 core electrons (1 s2 ). The atomic number of copper is 29 with electronic configuration 1s2 2s2 p6 3s2 p6 d10 4s1 ). For neutral atoms, the number of valence electrons is equal to the atoms main group number. Valence shell electrons (or, more simply, the valence electrons) are the electrons in the highest-numbered shell, or valence shell, while core electrons are the electrons in lower-numbered shells. 
Atoms share electrons and form covalent bonds to satisfy the octet rule. This page titled 5.2: Covalent Bonds and the Periodic Table is shared under a CC BY-NC-SA 3.0 license and was authored, remixed, and/or curated by Anonymous. The atoms in group 6A make two covalent bonds. It is more complex to determine the number of valence electrons in some elements like copper. Covalent bonds are formed by two atoms sharing electrons. Similarly there are seven valence electrons in bromine (Br = 1s22s2p63s2p6d104s2p5). Study with Quizlet and memorize flashcards containing terms like Only valence electrons are involved in, Which energy shell are valence electrons located. Hence 3s is the valance shell and electrons in this shell are valence electrons of Mg. Using the variable n to represent the number of the valence electron shell, write the valence shell electron configuration for each group. There are three shells, out of that third shell consist of two electrons. The shape of the periodic table reflects the order in which electron shells and subshells fill with electrons. For example, Atomic number of magnesium is 12, therefore electronic configuration will be 1s22s2p63s2. Therefore valence electrons are responsible for different chemical and physical properties of an element. The outermost energy level in an atom is called as valence shell and electrons placed in this shell are known as valence electrons. These electrons are mainly in the bond formation and chemical reaction with other atoms. First electrons filled in low energy levels and then move to higher energy level. The length of a period is determined by the number of electrons needed to fill the electron sublevel. 
Periods are the horizontal rows of the periodic table. Valency & Valence Electrons How to calculate valency of any element Chemistry Concept. Valence electrons are the electrons in the outermost shell, or energy level, of an atom. In an atom, electrons filled in different levels according to their energies. For d-block elements, the group numbers is (n-1) the number of electrons in the subshell plus the number of electrons in the valence shell. 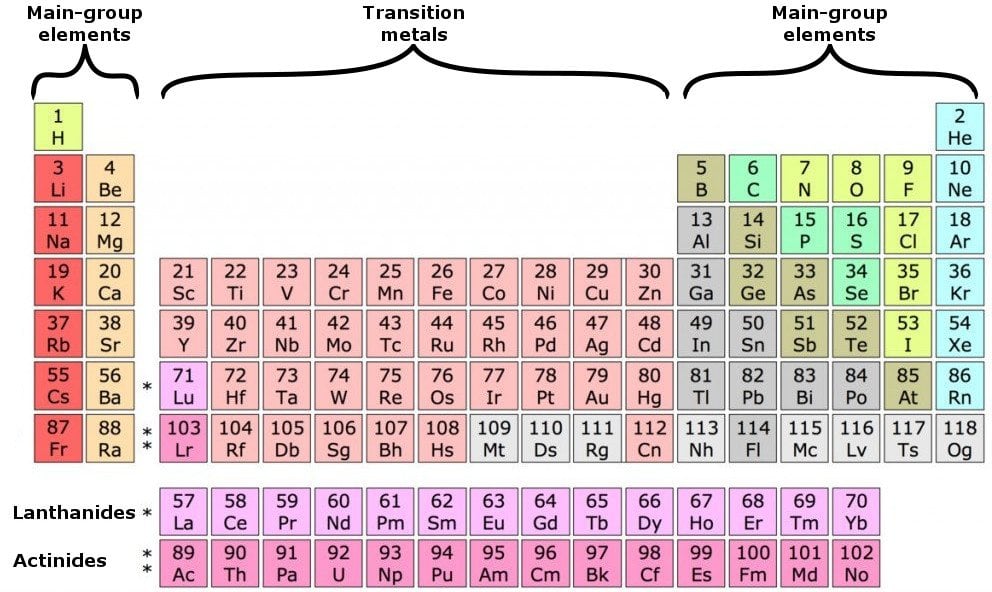 
Valence Electrons Configuration and the Periodic Table
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
